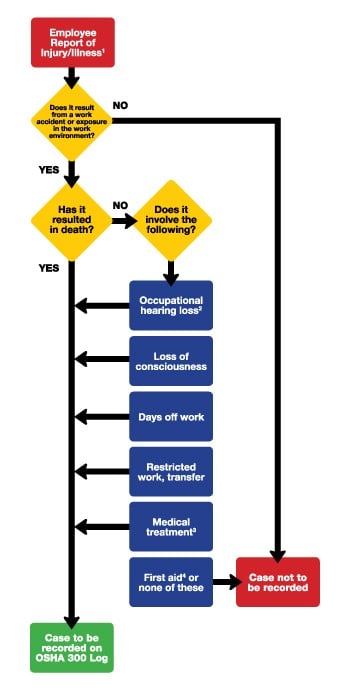
Employers are responsible for recording all work-related injuries and illnesses. If you are unable to determine if an injury or illness is recordable after you have completed the investigation, and evaluated all available documents, it is recommended that you contact the OSHA area office nearest you.
Read MoreEnvironmental Health and Safety Blog | EHSWire
Topics: OSHA, OSHA Recordkeeping regulation (29 CFR 1904), severe work-related injury and fatality reporting, OSHA Compliance, OSHA Hazard Communication Standard, reporting, Medical Records, OSHA Injury and Illness Recordkeeping and Reportin
The Occupational Safety and Health Administration’s (OSHA) recordkeeping requirements have been in place since 1971 (29 Code of Federal Regulations CFR Part 1904). The requirements were updated in 2002 to make it easier for employers to comply. OSHA has again updated the recordkeeping rule for 2015 to include two key changes.
Read MoreTopics: OSHA, OSHA Recordkeeping regulation (29 CFR 1904), health and safety, OSHA Compliance, Occupational Health, health hazards, occupational health and safety, reporting, Medical Records, OSHA Injury and Illness Recordkeeping and Reportin, OSHA 300A Annual Recordkeeping Summary Form, incident investigation
Emilcott TSCA Resource Center Expands with More Info and Options
Posted by Shivi Kakar
TSCA questions are pouring in and we are responding. To ensure that the information is available in a reasonable (and non-overwhelming) way, Emilcott has created a TSCA resource section of our web site for centralizing all kinds of intelligence, notifications, links, and summaries about Toxic Substance Control Act (TSCA) 2012 Chemical Data Reporting (CDR).
All these pages (just click on the headers) are available from the Emilcott Home page but we suggest you bookmark the pages that are most relevant and dont forget to register for our December 6th Free Webinar!
Emilcott TSCA Resource Center
The TSCA Resource Center has moved from the Emilcott home page to a new page lots of information including all our TSCA-related EHSwire blogs (CDR and IUR) and EPA Chemical Data Reporting Links, EPA New Chemicals Links, and EPA Import/Export Links. This page will stay updated so that you have a one-stop location for all TSCA information. If you have specific issues youd like to see addressed here, please let us know.
Topics: Compliance, TSCA & R.E.A.C.H., TSCA, CDR, Toxic Substance Control Act, IUR, violation, regulations, questions, filing, reporting, Chemical Data Reporting, Form U
What You Need to Know: TSCA 2012 CDR Form U Submission
Posted by Shivi Kakar
If you are a foreign or domestic business in the US who is either a chemical importer (resells for use in blending, repackaging) or chemical manufacturer (make new chemicals out of chemicals purchased from others with the exception for pharmaceutical companies), this update is about mandatory compliance with the EPAs Toxic Substances Control Act (TSCA), specifically filing the 2012 TSCA Form U Chemical Data Report.
Information about the new TSCA CDR Form U reporting tool is rolling in from the EPA. On September 23 rd the Agency hosted an hour-long webinar in which the 2012 CDR reporting requirements were reviewed and the use of the electronic Form U reporting tool (e-CDRweb) was demonstrated. If you missed the webinar or need a rewind, both the presentation materials and recorded webinar have been posted by the EPA at IUR/CDR About Submissions.
Webinar Take Aways
- The Agency is emphasizing two new reporting requirements:
- The standard of known to or reasonably ascertainable by for processing and use information (formerly readily obtainable)
- The upfront Confidential Business Information (CBI) substantiation
- Both the company that contracts for the manufacture AND the toll manufacturer are now considered to be the co-manufacturers of that chemical substance
- The e-CDRweb tool is designed for joint reporting and has very specific requirements for supplier-EPA communication. Joint reporting is specifically for those instances where a supplier will not disclose the specific chemical name (or TSCA accession number) of a chemical substance or a reactant used to manufacture the TSCA chemical substance because the name is claimed confidential.
- Registration with the EPAs Central Data Exchange (CDX) is required prior to accessing e-CDRweb.
- CDX registration requires the completion of an electronic signature agreement (ESA) form that foreign suppliers must submit by mail.
- CDX registration for e-CDRweb will be available on November 1, 2011.
- CDX registration is a multi-part process.
- The EPA will be providing support for use of the new e-CDRweb tool.
- A training webinar is tentatively scheduled for November 2011.
- Comprehensive instructions for the 2012 TSCA Chemical Data Reporting are now online.
To summarize, the e-CDRweb tool looks like it is a much friendlier submittal tool than its predecessor, e-IURweb. The gotcha will be in the preparation of the materials so that the submittal process is easy. Like many other federal filings, understanding what to submit and why may be much more complicated than the actual filing process. The requirements that changed from the 2006 to 2012 range wildly from subtle and minor to extensive and complex.
Did you participate in the EPAs e-CDRweb webinar? What did you think? Are there any particular gotchas that caught your eye?
If you need guidance with what data you should for the EPA TSCA 2012 CDR Submission, please contact Emilcott for help
- You can also subscribe to our free TSCA e-newsletter which delivers TSCA-related information right to your mailbox.
- Want more info? Enroll for our free Dec webinar by sending an email to pkaufmann@emilcott.com.
If you have any questions, feel free to post them in the comments section and we will respond quickly.
Topics: EPA, Compliance, TSCA & R.E.A.C.H., TSCA, Toxic Substance Control Act, submission, IUR, reporting, Form U, cdx, regulation, eCDRweb, central data exchange
Paula Kaufmann, CIH
As an occupational and safety professional, Ive noticed that the Occupational Safety and Health Administration (OSHA) has been busy over the last few weeks! The following is a summary of highlights of interest to Emilcott clients. Did any of these catch your attention?
Highlight #1: Up-to-Date OSHA Standards
Announcement of a final rule to help keep OSHA standards up-to-date and better enable employers to comply with their regulatory obligation. The concept should allow OSHA to easily remove outdated requirements, streamline and simplify standards without reducing employee protection. The rule is to be published soon in the Federal Register: OSHA Standards Improvement Project-Phase III final rule.
Benefit to employers: OSHA estimates that the final rule will result in annual cost savings to employers exceeding $43 million. Now thats an improvement to cheer about!
In the news release, OSHA stated that there will not be any NEW requirements set by this rule, so employers will be able to comply with it immediately. (However, it seems that there will be modifications...Emilcott will be keeping a lookout for those and post an update below or as a new EHSWire post.) Here are some examples listed in the news release on this rule:
- Respiratory Protection
- Aligning air cylinder testing requirements for self-contained breathing apparatuses with U.S. Department of Transportation regulations
- Clarifying that the provisions of Appendix D, which contains information for employees using respirators when not required under the standard, are mandatory if the employee chooses to use a respirator.
- Sanitation
- Defining potable water to meet the current Environmental Protection Agency
- Access to Exposure and Medical Records
- Deleting a number of requirements for employers to transmit exposure and medical records to NIOSH
- Slings
- Requiring that employers use only slings marked with manufacturers' loading information
Highlight #2: OSHA Injury and Illness Logs - Musculoskeletal Disorders (MSD)
Reopening the public record on proposed record-keeping rule to add work-related musculoskeletal disorders column. This keeps popping up!
- In January of 2010, OSHA proposed to revise its Occupational Injury and Illness Recording and Reporting Requirements regulation to restore a column to the OSHA 300 log that employers would have to check if an incident they already have recorded under existing rules is an MSD.
- On January 25, 2011, OSHA withdrew this proposed revision.
- On May 17, 2011, OSHA reopens the public record on a proposed rule.
Highlight #3: A Survey of Private Sector Employees
Launch of a targeted employer survey to collect information that would improve the development of future rules, compliance assistance and outreach efforts.
- The survey will be sent to private sector employers of all sizes and across all industries under OSHA's jurisdiction. Questions include whether respondents already have a safety management system, whether they perform annual inspections, who manages safety at their establishments and what kinds of hazards they encounter at their facilities. Participation in the survey is voluntary.
Highlight #4: Fall Protection for Residential Construction Workers
Online presentation about fall protection specifically designed for residential construction workers. (This is really great as residential construction crews frequently overlook safety just look at all the roofers walking around the top of your neighborhood homes!)
- The presentation describes safety methods for preventing injuries and deaths from falls, and explains techniques currently used by employers during various stages of construction. These techniques involve the use of conventional fall protection systems including safety nets, guardrails, and personal fall arrest systems such as body harnesses, lanyards and lifelines.
- The presentation is among a series of guidance materials on OSHA's Residential Fall Protection Web page.
On a personal note, my son is currently volunteering as a roofer on a Habitat for Humanity home construction site he informs me that he is wearing fall protection and the roof has anchor points!
So, can you tell that Emilcott is pretty excited about these changes? Instead of putting the onus on employers to become more aware of OSHA, OSHA is streamlining existing rules to match other government agencies (radical!), listening to employers before leaping into new regulations, and looking at alternative messaging techniques to market segments that frequently fall in the cracks.
If youre interested in whats happening at OSHA, just take a look at the loooooong list of May press releases Are there any highlights that you think important to you or American businesses? Any predictions for June?
Topics: OSHA, General Industry H&S, OSHA Compliance, General EHS, Construction H&S, Compliance, worker safety, reporting, regulation, construction, fall protection, federal register, log, standards, musculoskeletal disorder
We have some news on this years TSCA IUR Form U submission. Well, it really isnt information about the requirements but we do know that this years Form U submission period will not be June 1 to September 30. It will most likely be later this year. So, we all can move that task to another segment of the calendar year!
More Information
On May 11, 2011, the EPA issued a Federal Register Notice amending the Toxic Substances Control Act (TSCA) Inventory Update Reporting (IUR) regulations by delaying the June 1 to September 30 submission period for the 2011 Form U reports. The notice indicated that this delay will not alter the timing of subsequent submission periods (e.g., the submission period from June 1, 2016 to September 30, 2016). This is an interesting statement as one of the changes included in the proposed IUR Rule is a change of the reporting period cycle to every four (4) years from the current five (5) year cycle.
The EPA is delaying the submission period because the proposed IUR modifications rule has not yet been finalized. EPA expects to have the final version of the changes to the IUR reporting requirements in the near future. The revised 2011 submission period will be announced with the publication of the final IUR modification rule.
How does this delay what the EPA rule refers to as a suspension affect what needs to be done for the 2010 reporting period? It seems that the EPA will mandate a new submission period but it is not clear when this will be during 2011.
- We are assuming that the reporting period will remain as the 2010 calendar year.
- Our next assumption, or guess, is that the Form U submission period will shift to September 1 to December 31, but that will require that the final rule on the IUR modifications be published very soon.
Food for Thought
As recently as March 4, 2011, representatives from the American Petroleum Institute (API) met with the EPA presenting concerns about several aspects of the proposed IUR modifications rule. One topic the API presented was that when the last set of revisions of the IUR was finalized in 2003 with the next reporting period was extended by one year shifting from 2004 to 2005 with Form U submission in 2006. During 2004 and 2005, the EPA held many workshops and issued clarification and guidance documents.
And, for now, we wait for the Final Rule and hope that the data we have all collected for the 2010 reporting period will be adequate. Emilcott's recommendations for what to do while we wait are in my January blog: TSCA IUR Update What Are the Changes ?. Essentially, we are advising our clients to proceed with the collection of 2010 inventory data with a threshold of 25,000 lbs. Here are a couple of items to keep on your radar:
- Be sure your list of manufactured chemicals is complete. Your list should be based on all chemical processes and imported materials received at the site and not just on the products.
- When calculating individual substance volumes include imported mixtures with those manufactured at the site aggregating all mixtures containing that substance.
...And, Emilcott will continue to keep you posted!
What to do if you need help or have questions?
If you need assistance related to the TSCA New Chemicals regulatory requirements or the potential changes due to the Inventory Update Reporting Rule, Emilcott can guide you through the reporting. We can also help you navigate the maze of reporting a potential Form U violation from prior filing years to the EPA (See http://www.emilcott.com/services/svcenvcompliance.asp).
As more information becomes available from the EPA regarding the IUR and as testing of the electronic tool begins, Emilcott will keep you up-to-date via EHSWire and our "Regulatory Updates" Newsletter.
Please give me a call at 1-800-886-3645 or write a comment below if you have any questions or additional information to contribute.
Topics: Emilcott, health and safety, General EHS, EPA, Compliance, TSCA & R.E.A.C.H., TSCA, Toxic Substance Control Act, reporting, regulation, chemicals
In the August 13, 2010 TSCA Inventory Update Reporting Modifications - Proposed Rule, the EPA anticipated the promulgation of the final rule by the Spring of 2011. Spring arrived 3 weeks ago, and the final IUR Reporting Modification rule has not been published. As such, the reporting modifications and specific reporting period have not been finalized. In addition, the Agency has not released a test version of the revised Form U electronic reporting software. It is possible that the Agency will change the proposed 2011 submission period (June 1 September 30) to another 4month period later in 2011.
EPA's intended final rule was sent to the White House Office of Management and Budget (OMB) on January 20, 2011. Since the beginning of February, OMB has held meetings with the American Chemistry Council, the Society of Chemical Manufacturers and Affiliates Inc. (SOCMA), the Small Business Administration, the National Mining Association, and associations that represent companies that must comply with the regulations.
So -- once again, the question is now what to do?
As mentioned in my January blog TSCA IUR Update What Are the Changes ? we are advising Emilcott clients to proceed with the collection of 2010 inventory data with a threshold of 25,000 lbs. Here are a couple of items to keep on your radar:
- Be sure your list of manufactured chemicals is complete. Your list should be based on all chemical processes and imported materials received at the site and not just on the products.
- When calculating individual substance volumes include imported mixtures with those manufactured at the site aggregating all mixtures containing that substance.
Additional data that may be needed for the 2011 reporting are listed below. Depending upon how you gather your information, you may want to request this along with the import or manufacturing volume information.
- Production volumes at or above 25,000 lbs directly exported and not domestically processed or used.
- All quantities of substances subject to rules and orders in the following sections:
- Section 5(a)(2) Significant New Use Rules (SNURs)
- Section 5(b)(4) Chemicals of concern to EPA
- Section 6 Prohibitions for chemicals with unreasonable risks
- Section 5(e) Requirements or restrictions on chemical production or use
- Section 5(f) Chemical with an unreasonable risk
What to do if you need help?
If you need assistance related to the TSCA New Chemicals regulatory requirements or the potential changes due to the Inventory Update Reporting Rule, Emilcott can guide you through the reporting. We can also help you navigate the maze of reporting a potential Form U violation from prior filing years to the EPA (See http://www.emilcott.com/services/svcenvcompliance.asp). As more information becomes available from the EPA regarding the IUR and as testing of the electronic tool begins, Emilcott will keep you up-to-date via EHSWire and our Regulatory Updates Newsletter. If you have any TSCA IUR questions or concerns, feel free to contact Emilcott or post your question below!
Topics: EPA, Compliance, TSCA & R.E.A.C.H., TSCA, Toxic Substance Control Act, reporting, regulation, chemicals
Regulatory Submissions & Postings Reminder (January thru April 2011)
Posted by Shivi Kakar
Here is a handy table we recently created for our clients -- a gentle reminder to get organized! Even if you miss a deadline, it's better to start playing catchup as soon as you find out that you are not in compliance.
Want to stay informed? Emilcott publishes a timely email reminder, "EHS Regulatory Submissions", 3x/year to keep our clients informed about upcoming deadlines. If you'd like to subscribe to that newsletter, just go to http://www.emilcott.com/subscribe.asp. If you need help with your Regulatory Submissions, contact Emilcott and ask for either an EHS or Hazardous Materials/Waste consultant.
Quick Reference Guide to Regulations and Submissions (Jan-Apr 2011)
| Regulation | Submission | Frequency | SUBMISSION DATE |
| EPA TSCA New Chemicals | TSCA Polymer Exemption Report | Annual | January 31 |
| EPA Greenhouse Gas Reporting | Certificates of Representation | Registration | January 31 |
| EPA Greenhouse Gas Reporting | GHG Reports | Annual | March 31 |
| OSHA Recordkeeping & Reporting Occupational Injuries & Illnesses | OSHA Injury and Illness Log Summary Form 300A | Annual | Post Feb 1 thru April 30 |
| NJ Emissions Statement Rule | Emission StatementNon-applicability Report | As Warranted | Feb 1 |
| Emergency Planning and Community Right-to-Know Act (EPCRA) Section 312 | Community Right-to-Know (CRTK) Survey and Tier I or Tier II Inventory Form | Annual | March 1 |
| NPDES Stormwater Program | Annual Certification | Annual | Varies by State |
EPA TSCA New Chemicals
Anyone who imports or manufactures a new polymer in 2010 that met the TSCA Exemption Criteria must submit a TSCA Polymer Exemption Report of manufacture or import by (postmarked) January 31 of the year subsequent to initial manufacture. The notice must include:
Topics: OSHA, health and safety, General Industry H&S, OSHA Compliance, General EHS, Construction H&S, EPA, Hazardous Waste Management, Compliance, worker safety, Occupational Health, Occupational Safety, TSCA & R.E.A.C.H., TSCA, reporting, Form 300, Greenhouse Gas Reporting
OSHA Recordkeeping 300A Its For You and the Rest of the Country!
Posted by Shivi Kakar
Did you know that the OSHA Illness and Injury Summary Log, 300A, is used for more than just recordkeeping at your site? By documenting your companys illness and injuries properly, you shape OSHAs future initiatives! Specifically, OSHA Summary 300A Forms are gathered by the OSHA Data Initiative (ODI) to help direct OSHA programs and measure its own performance.
How does OSHA get this information?
OSHA gets these data from two sources:
- As part of an annual survey, the Bureau of Labor Statistics (BLS) sends injury and illness survey forms to randomly selected employers and uses the information to create the Nation's occupational injury and illness statistics.
- The OSHA Data Initiative mails its annual survey (in June) that collects data on injuries and acute illnesses attributable to work-related activities in private-sector industries from approximately 80,000 establishments in selected high hazard industries. In 2010, OSHA also collected this information from approximately 20,000 establishments in the construction industry in addition to the non-construction establishments. The Agency uses these data to calculate establishment-specific injury/illness rates, and in combination with other data sources, to target enforcement and compliance assistance activities. Traditionally, OSHA collects data from the establishments that meet the following categories, but as we saw in 2010, OSHA can expand these criteria.
- Non-construction industries with 40 or more employees are chosen randomly
- Non-respondents in the previous collection year
- Site with an inspection or consultation visit for performance measurement
- DART rate (days away from work, restriction or transfer) of 7.0 or higher in previous data collection
Does the data really help OSHA?
Now that OSHA has the data from BLS and the ODI, the Agency uses the information to
- Calculate and establish specific injury and illness incidence rates
- Develop targeted intervention programs (i.e., inspections and enforcement action)
- Assist inspectors so that they can direct their efforts to the higher incidents hazards that are hurting workers.
- Measure the success of agency efforts to reduce the number of workplace injuries and illnesses in select high-hazard industries
- Provide the base data for the BLS Annual Survey of Occupational Injuries and Illnesses, the Nation's primary source of occupational injury and illness data.
What is your role?
Be a savvy and educated reporter of your companys illness and injury information. Understanding the OSHA 300 log reporting requirements will ensure an accurate portrayal of worker health and safety as well as maintaining OSHA compliance. Can you answer the following questions?
- How do I complete the OSHA 300 Log and Form 301?
- Am I required to post an OSHA Form 300A ? How do I know if I am exempt?
- What is classified as a work-related illness or injury?
- How do I fill the forms in correctly without over-reporting?
- Do I have to fill in the form if I have no recordable injuries or illnesses in the previous year? What are the rules for posting?
- Once the form is filled in and submitted, if requested by BLS or OSHA, are there other legal requirements I should know?
Not convinced that recordkeeping is important?
Besides providing a visible record of worker safety benchmarks and improvements (or worse, tragedies and reversals), establishments that are requested to but fail to submit a completed data collection form may be subject to OSHA enforcement actions, including the issuance of a citation and assessment of penalties! So, take the time understand OSHAs reporting requirements and implement them correctly it affects your company and the nations workers.
If you need assistance with OSHA recordkeeping, Emilcott offers a variety of ways to help your business stay in compliance from a webinar-based course that outlines the rules and regulations to the development of complete health and safety plans. Or, if you have an OSHA recordkeeping question, just ask us!
Topics: Emilcott, OSHA, health and safety, General Industry H&S, OSHA Compliance, General EHS, Construction H&S, Compliance, worker safety, Occupational Health, Occupational Safety, Webinar, reporting, regulation, Medical Records, BLS, Bureau of Labor Statistics
2010 Holiday Planning Includes 2011 EPA Submission Deadlines
Posted by Shivi Kakar
Another calendar year is drawing to a close; where does the time go? As I plan my own holiday celebrations and commitments, environmental professionals like me have another type of planning to keep in mind. With the start of each new year, we face regulatory submission deadlines reporting data from the past year including Submission of the EPA Community Right to Know (CRTK) Survey -- a Federal act with each state managing their own program due March 1 st and EPA Toxics Release Inventory (TRI) due July 1 st.
Just like Christmas shopping, the compilation and reporting process is less stressful and yields better results if I begin early and develop a strategy with deadlines in mind. As such, here is my personal January 1 st kick-off list that should make the time-consuming process of CRTK and TRI reporting easier to handle.
1) Start requesting and gathering all the information needed for these submittals.
- 2010 purchasing records of the chemicals you are reporting
- 2010 production logs where these chemicals are used
- 2010 waste information
- 2010 recycling information for any reported chemicals that were recycled
- 2010 air emission inventory
2) Develop and write down a comprehensive set of due dates so that you have time to review information as it comes in. If the requested data is late, have a plan to follow up or find another source because the deadline is not going to change!
3) Review the rules early to avoid unpleasant surprises. For example, The Environmental Protection Agency (EPA) finalized a rule effective November 10, 2010 which added 16 chemicals to the list of TRI reportable chemicals. To ensure that you are reporting what you need to report, check the TRI database on the EPA website: http://www.epa.gov/tri/trichemicals/index.htm.
4) Allow time for anomalies and additional fact-finding. As Charles Peruffo described in a recent EHSWire blog about filing the NJ PPA, reported amounts from different sources may not match. If you find that is the case, its your job to figure out why and that always adds more time to the already challenging process.
Emilcotts clients depend on our environmental knowledge and organizational capabilities to gather the required information on time and give them fair warning if there is trouble ahead. My best advice for successful reporting-dont wait until the last minute. Much like shopping for Christmas on Dec 24th, waiting until February to gather the information for the CRTK or starting in June for the TRI will be stressful and could result in costly errors. So, what am I doing today? Like Santa, Im checking my own list twice!
Have you been meeting the CRTK and TRI deadline? If yes, can you offer additional advice or do you have particular steps that you take to get the submission process rolling?
Topics: General EHS, EPA, Hazardous Waste Management, HazCom, Hazardous Materials, reporting, Toxics Release Inventory, emissions, Community Right to Know, haz waste, TRI, CRTK, chemical